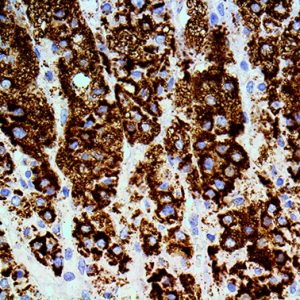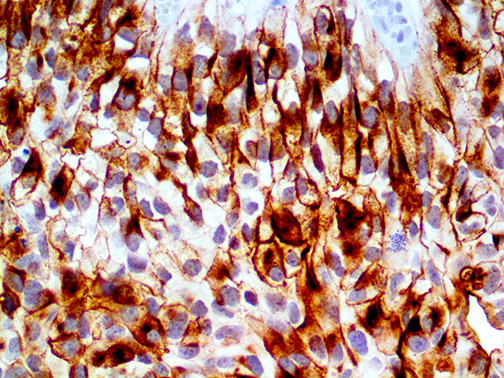
IHC of Uroplakin III on an FFPE Bladder TCC Tissue
Uroplakin III, RMab
| Availability |
| |||||||||||||||||||||||||||||||||||
| Note: For concentrated antibodies, please centrifuge prior to use to ensure recovery of all product. | ||||||||||||||||||||||||||||||||||||
Uroplakins (UPs) are a family of transmembrane proteins (UPs Ia, Ib, II and III) that are specific differentation products of urothelial cells. In non-neoplastic mammalian urothelium, UPs are expressed in the luminal surface plasmalemma of superficial (umbrella) cells, forming complexes of 16-nm crystalline particles. Uroplakin
III is expressed in urothelial carcinomas, whereas many non urothelial carcinomas were UPIII-negative. Recent studies have shown that UP expression might reflect the malignant potential of urothelial cancer cells as well as being cytodifferential markers of urothelial cells.
UPIII expression is strongly associated with lower tumor grades and lack of UPIII expression in urothelial tumors of the upper urinary tract is associated with much higher rates of metastases. Five-year specific survival is much worse for UPIII negative tumors (54%) than for UPIII positive tumors (100%). Apparently UPIII expression is a better indicator fo the malignant potential of the tumor than the grade of the tumor.
| Intended Use | For In Vitro Diagnostic Use |
|---|---|
| Antibody Type | Rabbit Monoclonal |
| Clone | EP321 |
| Isotype | IgG |
| Reactivity | Paraffin, Frozen |
| Localization | Cytoplasmic, Membranous |
| Control | Bladder, Bladder Carcinoma |
Uroplakin III is a rabbit monoclonal antibody derived from cell culture supernatant that is concentrated, dialyzed, filter sterilized and diluted in buffer pH 7.5, containing BSA and sodium azide as a preservative.
- Badalament RA, et al. J Urol. 1990; 144:859-63
- Hall MC, et al. Urology. 1998; 52:594-601
- Ohtsuka Y, et al. BJU Int. 2006 Jun; 97(6):1322-6
- Oldsburgh J, et al. J Pathol. 2003; 199:41-9
- U.S. Department of Health and Human Services: Centers for Disease Control and Prevention. Guidelines for Safe Work Practices in Human and Animal Medical Diagnostic Laboratories. Supplement / Vol. 61, January 6, 2012.