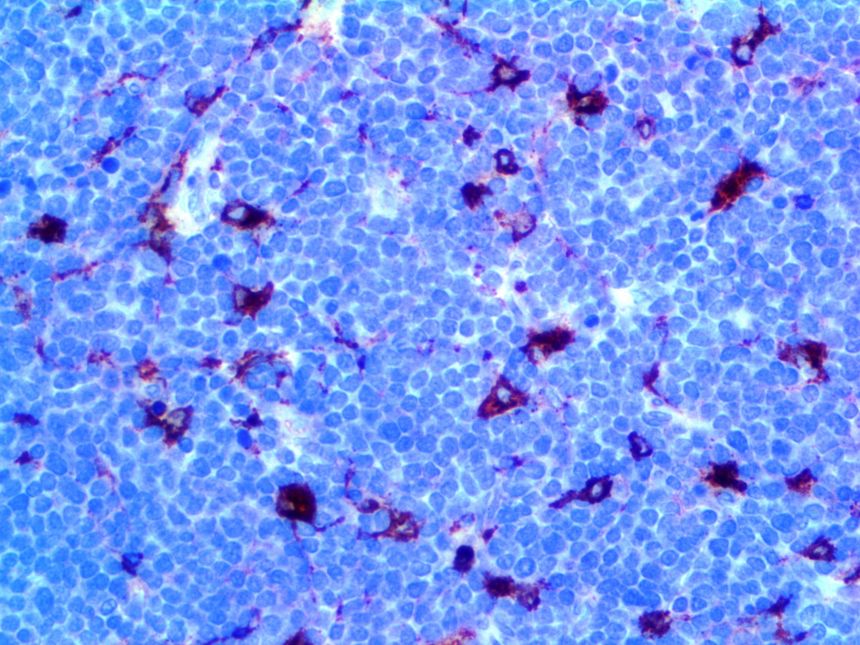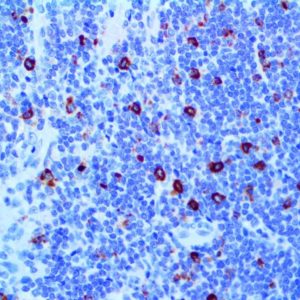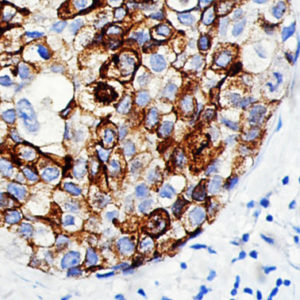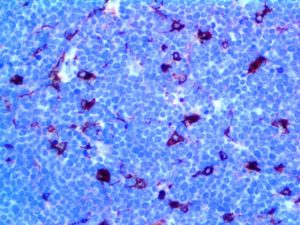
IHC of MMP-9 on an FFPE Lymphoblastic Lymphoma Tissue
| Intended Use | For In Vitro Diagnostic Use | |||||||||||||||||||||||||||||||||||
| Summary and Explanation | The matrix metalloproteinases (MMPs) are responsible for degradation of the extracellular matrix. The MMPs and their specific tissue inhibitor metalloproteinases (TIMP) have been associated with tumor cell invasion and metastasis in a number of adult tumors. MMP-9, also designated as 92-kDa Type IV Collagenase or gelatinase B, is a member of MMPs, which is produced as a 92- kDa pro-enzyme by neutrophils, macrophages, mast cells and stromal cells, as a normal constituent and released into the extracellular environment after activation in inflammatory tissues. MMP-9 may be involved in the development of several human malignancies, as degradation of collagen IV in basement membrane and extracellular matrix facilitates tumor progression, including invasion, metastasis, growth and angiogenesis. The expression levels of MMP-9 in tumors are elevated compared with the corresponding normal tissues in a variety of cancer types, including breast, colon, gastric and nasopharyngeal cancers. MMP-9 may play an important role in angiogenesis and neovascularization. For example, MMP9 appears to be involved in the remodeling associated with malignant glioma neovascularization. Increased expression has been seen in a metastatic mammary cancer cell line. | |||||||||||||||||||||||||||||||||||
| Antibody Type | Rabbitt Monoclonal | Clone | EP-127 | |||||||||||||||||||||||||||||||||
| Isotype | IgG | Reactivity | Paraffin, Frozen | |||||||||||||||||||||||||||||||||
| Localization | Cytoplasmic | Control |
| |||||||||||||||||||||||||||||||||
| Presentation | MMP-9 is a rabbit monoclonal antibody derived from cell culture supernatant thatis concentrated, dialyzed, filter sterilized and diluted in buffer pH 7.5, containing BSA and sodium azide as a preservative. | |||||||||||||||||||||||||||||||||||
| Availability |
| |||||||||||||||||||||||||||||||||||
| Note: For concentrated antibodies, please centrifuge prior to use to ensure recovery of all product. | ||||||||||||||||||||||||||||||||||||