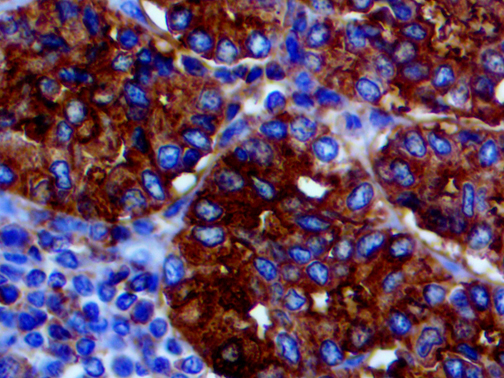
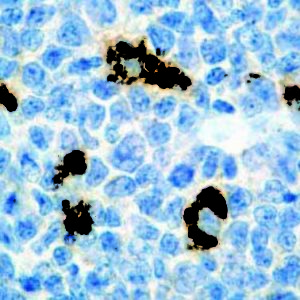
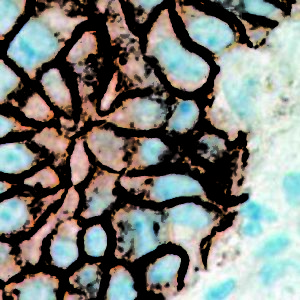
IHC of CA19-9 on an FFPE Salivary Gland Tissue
| Intended Use | For In Vitro Diagnostic Use | |||||||||||||||||||||||||||||||||||
| Summary and Explanation | CA19-9 (carbohydrate antigen 19-9 or sialylated Lewis (a) antigen) is a blood test from the tumor marker category. It was discovered in patients with Colon Cancer and Pancreatic Cancer in 1981. Increased levels of CA19-9 are also found in non-malignant conditions, such as Mirizzi’s Syndrome and diseases of the bile duct and liver. The main use of CA19-9 is to determine whether a pancreatic tumor is secreting it; if that is the case, then the levels should fall when the tumor is treated, and they may rise again if the disease recurs. CA19-9 antigen is highly expressed in Gastrointestinal (gastric, pancreatic, and colonic) Adenocarcinomas and salivary gland Mucoepidermoid Carcinomas. CA19-9 is usually not reactive with breast, kidney, and prostate Carcinomas, but is reactive with sialylated Lea-active pentasaccharide (sialylated lacto-N-fucopentaose II), which is enzymatically synthesized by sialylation of Type 1 carbohydrate chains. | |||||||||||||||||||||||||||||||||||
| Antibody Type | Mouse Monoclonal | Clone | 121SLE | |||||||||||||||||||||||||||||||||
| Isotype | IgM | Reactivity | Paraffin, Frozen | |||||||||||||||||||||||||||||||||
| Localization | Cytoplasmic | Control | Breast, Colon, Cervix, Pancreas, Pancreas Cancer, Breast Carcinoma, Colon Carcinoma, Transitional Cell Carcinoma, Ovarian Carcinoma Thyroid Carcinoma | |||||||||||||||||||||||||||||||||
| Presentation | CA19-9 is a mouse monoclonal antibody derived from cell culture supernatant that is concentrated, dialyzed, filter sterilized and diluted in buffer pH 7.5, containing BSA and sodium azide as a preservative. | |||||||||||||||||||||||||||||||||||
| Availability |
| |||||||||||||||||||||||||||||||||||
| Note: For concentrated antibodies, please centrifuge prior to use to ensure recovery of all product. | ||||||||||||||||||||||||||||||||||||